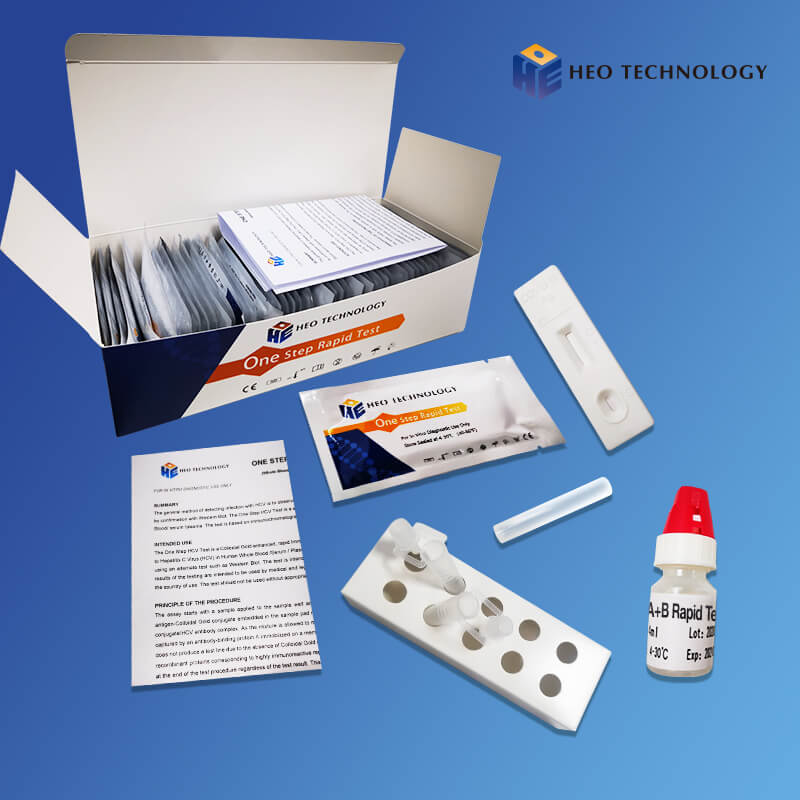
High definition Rapid Test Results - Dengue IgGIgM Test Device (Whole BloodSerumPlasma) – HEO
High definition Rapid Test Results - Dengue IgGIgM Test Device (Whole BloodSerumPlasma) – HEO Detail:
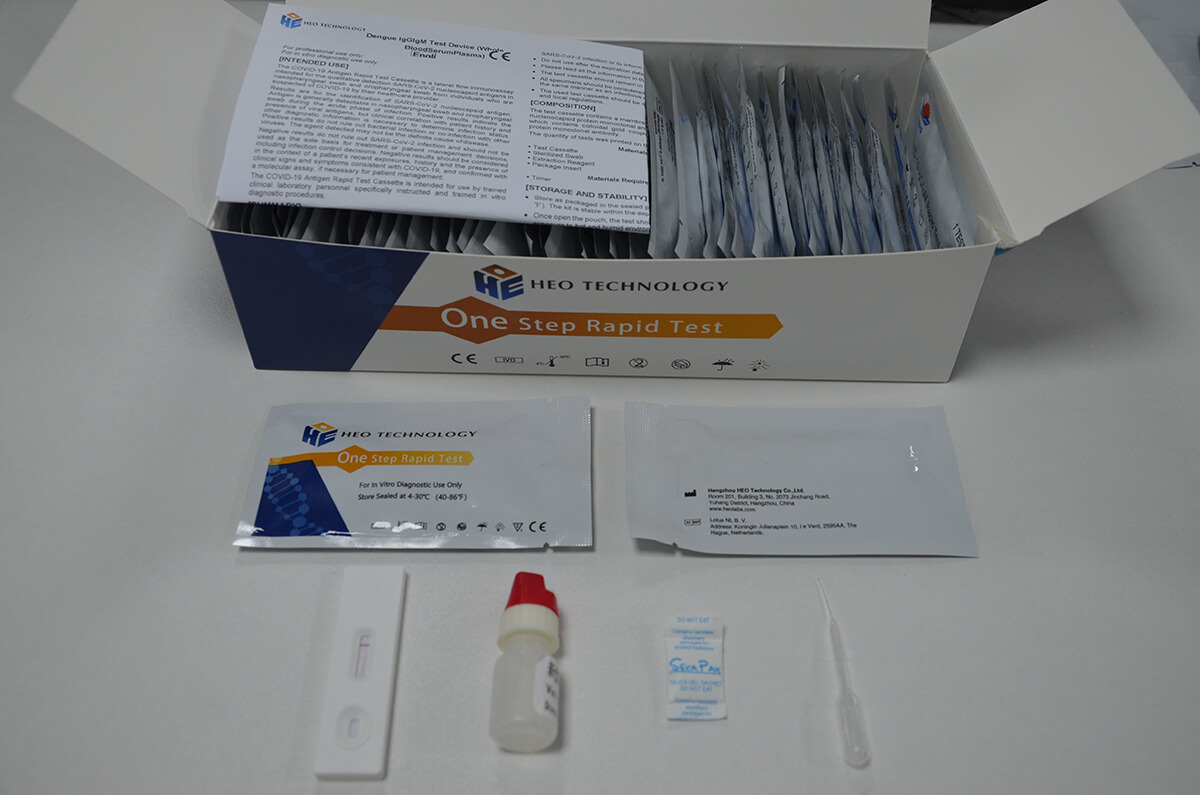
Dengue IgGIgM Test Device (Whole BloodSerumPlasma)
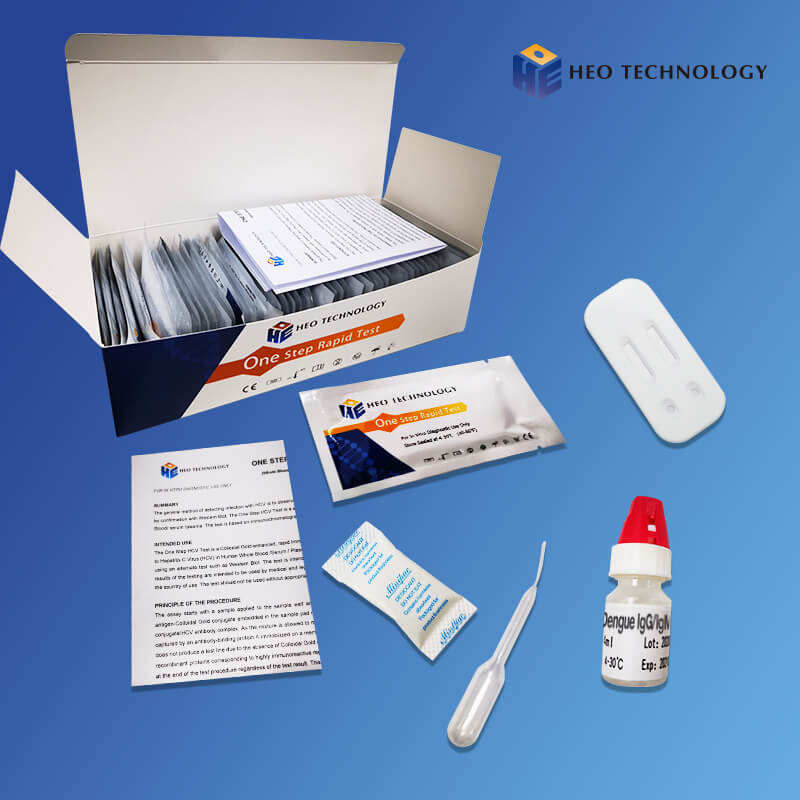



[INTENDED USE]
The Dengue IgG/IgM Rapid Test Cassette is a lateral flow chromatog- raphic immunoassay for the qualitative detection of antibodies (IgG and IgM) to dengue virus in human Whole Blood/Serum/Plasma. It provides an aid in the diagnosis of infection with Dengue viruses.
[SUMMARY]
Dengue fever is an acute vector-borne infectious disease caused by dengue virus transmitted by mosquitoes. Dengue virus infection can lead to recessive infection, dengue fever, dengue hemorrhagic fever, dengue hemorrhagic fever. Typical clinical manifestations of dengue fever include sudden onset, high fever, headache, severe muscle, bone and joint pain, skin rash, bleeding tendency, lymph node enlargement, decreased white blood cell count, thrombocytopenia and so on in some patients. This disease basically is in tropical and subtropical area popularity, because this disease is transmitted by Aides mosquito, reason popularity has certain seasonally, be in every year commonly in May ~ November, peak is in July ~ September. In the new epidemic area, the population is generally susceptible, but the incidence is mainly adult, in the endemic area, the incidence is mainly children.
[PRINCIPLE]
The Dengue IgG/IgM Rapid Test Cassette is a qualitative membrane strip based immunoassay for the detection of dengue virus antibodies (IgG and IgM) in human Whole Blood/Serum/Plasma. The test cassette consists of: 1) a burgundy colored conjugate pad containing dengue recombinant envelope antigens conjugated with Colloid gold (dengue conjugates), 2) a nitrocellulose membrane strip containing two test lines (IgG and IgM lines) and a control line (C line). The IgM line is pre-coated with the Mouse anti-Human IgM antibody, IgG line is coated with Mouse anti-Human IgG antibody. When an adequate volume of test specimen is dispensed into the sample well of the test cassette, the specimen migrates by capillary action across the cassette. IgM anti-dengue if present in the specimen will bind to the dengue conjugates. The immunocomplex is then captured by the reagent coated on the IgM line, forming a burgundy colored IgM line, indicating a dengue IgM positive test result and suggesting a fresh infection. IgG anti-dengue, if present in the specimen, will bind to the dengue conjugates. The immunocomplex is then captured by the reagent pre-coated on the IgG band, forming a burgundy colored IgG line, indicating a dengue IgG positive test result and suggesting a recent or repeat infection. Absence of any T lines (IgG and IgM) suggests a negative result. To serve as a procedural control, a colored line will always appear at the control line region indicating that proper volume of specimen has been added and membrane wicking has occurred.
[STORAGE AND STABILITY]
Store as packaged in the sealed pouch at temperature (4-30℃ or 40-86℉). The kit is stable within the expiration date printed on the labeling.
Once open the pouch, the test should be used within one hour. Prolonged exposure to hot and humid environment will cause product deterioration.
The LOT and the expiration date were printed on the labeling.
[SPECIMEN]
The test can be used to test Whole Blood/Serum/Plasma specimens.
To collect whole blood, serum or plasma specimens following regular clinical laboratory procedures.
Separate serum or plasma from blood as soon as possible to avoid hemolytic. Use only clear non-demolished specimens.
Store specimens at 2-8℃ (36-46℉) if not tested immediately. Store specimens at 2-8℃ up to 7 days. The specimens should be frozen at
-20℃ (-4℉) for longer storage. Do not freeze whole blood specimens.
Avoid multiple freeze-thaw cycles. Prior to testing, bring frozen specimens to room temperature slowly and mix gently. Specimens containing visible particulate matter should be clarified by centrifugation before testing.
Do not use samples demonstrating gross lineman, gross hemolytic or turbidity in order to avoid interference on result interpretation.
[TEST PROCEDURE]
Allow the test device and specimens to equilibrate to temperature (15-30℃ or 59-86℉) prior to testing.
1. Bring the pouch to room temperature before opening it. Remove the test cassette from the sealed pouch and use it as soon as possible.
2. Place the test cassette on a clean and level surface.
3. Hold the dropper vertically and transfer 1 drop of specimen (approximately 10μl) to the specimen well(S) of the test cassette, then add 2 drops of buffer (approximately 70μl) and start the timer. See illustration below.
4. Wait for the colored line(s) to appear. Read results at 15 minutes. Do not interpret the result after 20 minutes.
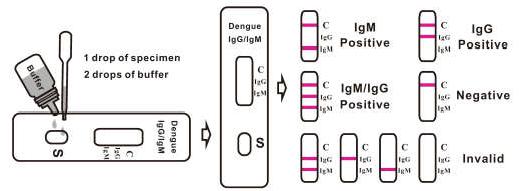
[INTERPRETATION OF RESULTS]
Positive: Control line and at least one test line appear on the membrane. The appearance of IgM test line indicates the presence of dengue specific IgM antibodies. The appearance of IgG test line indicates the presence of dengue specific IgG antibodies. And if both IgG and IgM line appear, it indicates that the presence of both dengue specific IgG and IgM antibodies. Negative: One colored line appears in the control region(C). No apparent colored line appears in the test line region.
Invalid: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test cassette. If the problem persists, discontinue using the test kit immediately and contact your local distributor.
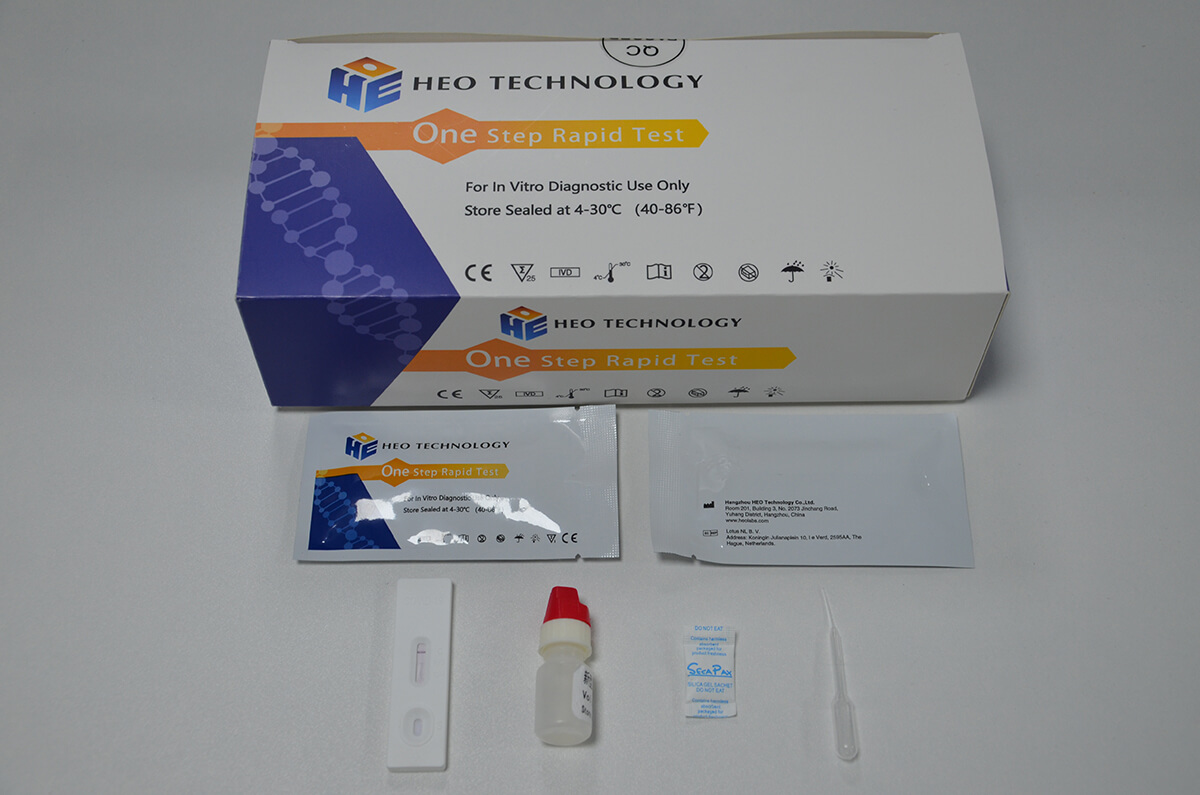
Product detail pictures:
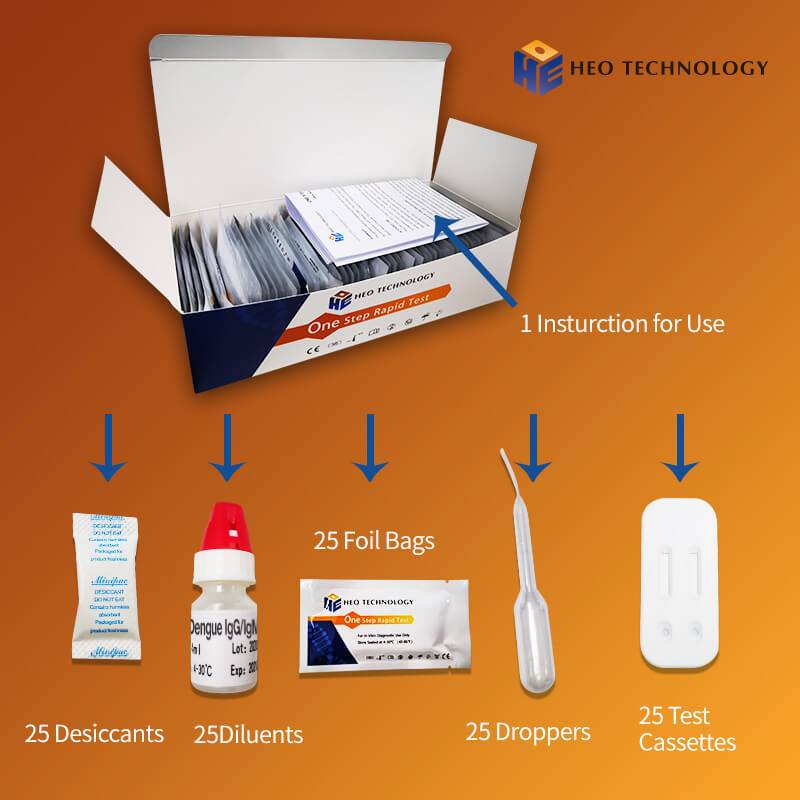

Related Product Guide:
Our personnel are always in the spirit of "continuous improvement and excellence", and with the superior quality products, favorable price and good after-sales services, we try to win every customer's trust for High definition Rapid Test Results - Dengue IgGIgM Test Device (Whole BloodSerumPlasma) – HEO , The product will supply to all over the world, such as: Myanmar, Lesotho, The Swiss, To achieve reciprocal advantages, our company is widely boosting our tactics of globalization in terms of communication with overseas customers, fast delivery, the best quality and long-term cooperation. Our company upholds the spirit of "innovation, harmony, team work and sharing, trails, pragmatic progress". Give us a chance and we will prove our capability. With your kind help, we believe that we can create a bright future with you together.
High Quality, High Efficiency, Creative and Integrity, worth having long-term cooperation! Looking forward to the future cooperation!